The world's phosphorus (P) reserves are almost peaking! Yet excess P is polluting our water resources! What is going on with the phosphorus cycle and what can be done about it?
Phosphorus, together with nitrogen, is the most important element in plant and animal nutrition. It is found in nature mostly in the form of the phosphate ions (PO43- and HPO42-). It is an important component of the nucleic acid molecules DNA and RNA, and of the cellular energy carrier adenosine tri-phosphate (ATP). P is also an important building block of bones and teeth, where it is found in the form of calcium phosphate.
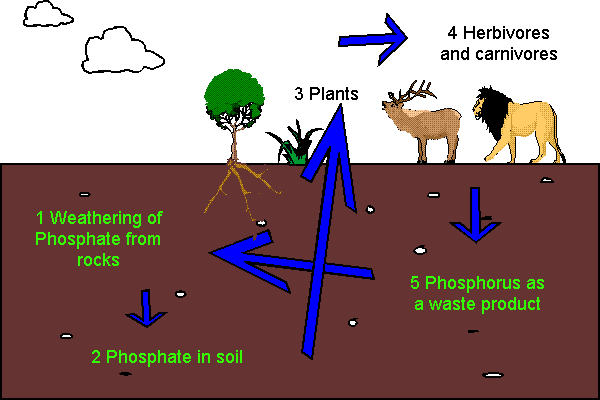
P cycle diagram created by Katie Corbin and sourced in Google images.
Most phosphates originate from sea sediments, brought to surface by geological uplift, and from rock weathering, over long periods of time. Plants absorb phosphates from the soil and use it in cellular processes. Phosphate taken up by plants may then be passed on to animals when the plant is consumed by herbivore that, in turn, may be consumed by carnivores. After death, animals and plants decay, and the phosphates are returned to the soil by way of bacterial decomposition. Runoff from the land may carry leached phosphate back to the ocean, where it eventually enters sediments and is reincorporated into rock, completing the P cycle. The industrial revolution and the resulting developments have resulted in a broken P cycle due to human interference, i.e. humans are mining and using more P than nature can recycle.
P is often a limiting nutrient for plant growth because only a small amount in phosphate form is available for plant uptake. For this reason, phosphate fertilizer is applied to fields to boost crop yields. In 2005, 14 million tonnes, of the 17.5 million tonnes P mined, was used in fertilizers production. However, there are concerns that supplies of P for fertilizers are limited (1). On the other hand, phosphate leached from farmland to the sea and other water bodies cause cyanobacterial blooms also known as eutrophication, resulting in poor water quality. This means it is quite obvious that wasted P should be recovered to avoid pollution of water courses and to avoid future shortages.
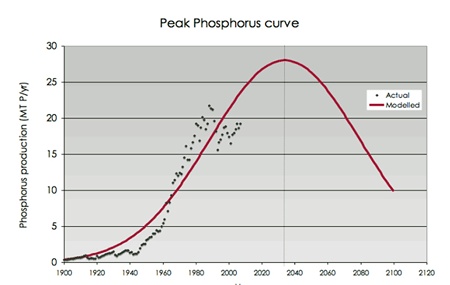
Diagram from Cordell et al. (2009)
'The Sustainable P Initiative' at Arizona State University is carrying out research tackling these two faces of P and James Elser, one of its co-founder, and Elena Bennett, from McGill University, said recently in an article in Nature (2) that we need to move quickly beyond academic discussions to creative policy solutions to the P problem; recycling programmes are urgently needed, they added.
Elser and Bennett pointed out that the solution is recycling: each person excretes about 1.2 g of P per day, which globally would amount to 3 million tonnes per year (20% of the annual P consumed worldwide). Currently only 10% of human excreted P is returned to agriculture. Urine-separating toilets are suggested, such as the NoMix toilet used in some European countries, which diverts the urine to a recycling centre. Elser and co-researchers, in an article in Chemosphere (3) also showed how plants have a number of adaptive responses to orthophosphate (Pi) limitation, a trait which should be used in plant genetic engineering to generate plants with enhanced Pi use efficiency.
The authors, in their Nature article, also called for the establishment of an international network policy research centre for nutrient sustainability, which would also include nitrogen and potassium; they even suggested the creation of ‘P emission markets’ similar to carbon markets. Is peaking P the next global crisis?
As a co-editor of the Soils & Fertilizers database at Cabi, I come across a lot of the latest literature on P and I have selected some of the latest records on P sustainability for further reading below, including a special issue of Chemosphere on ‘The Phosphorus Cycle’. The links are provided for the benefit of the CAB Abstracts and/or Environmental Impact susbscribers.
References
1 – Cordell, D.; Drangert, J. O.; White, S. (2009) The story of phosphorus: global food security and food for thought. Global Environmental Change, 2009, 19, 2, pp 292-305, many ref. http://www.agci.org/dB/PDFs/09S2_TCrews_StoryofP.pdf
2 – Elser, J. J.; Bennett, E. (2011) A broken biogeochemical cycle. Nature 478, pp 29-31.
3 – Gaxiola, R. A.; Edwards, M.; Elser, J. J. (2011) A transgenic approach to enhance phosphorus use efficiency in crops as part of a comprehensive strategy for sustainable agriculture. Chemosphere, 84(6):840-845.
- Edelstein, D. M.; Tonjes, D. J. (2012). Modeling an improvement in phosphorus utilization in tropical agriculture. Taylor & Francis, Philadelphia, USA, Journal of Sustainable Agriculture, 2012, 36, 1, pp 18-35, many ref.
- Vaccari, D. A.; Vaccari, D. A. (2011). Special Issue: The phosphorus cycle. Elsevier Ltd, Oxford, UK, Chemosphere, 2011, 84, 6, pp 735-853.
- Ragnarsdottir, K. V.; Sverdrup, H. U.; Koca, D. Anderson, S. P.(2011). Challenging the planetary boundaries I: Basic principles of an integrated model for phosphorous supply dynamics and global population size. Elsevier Ltd, Amsterdam, Netherlands, Applied Geochemistry, 2011, 26, Suppl. 1, pp S303-S306.
- Costanzi, R. N.; Frizzo, E.; Dombeck, D.; Colle, G.; Rosa, J. F. da; Maibuk, L. A. C.; Fernandes, M. da S. P. (2010). Yellow water reuse. Associação dos Engenheiros e Arquitetos de Ponta Grossa, Ponta Grossa, Brazil, Revista de Engenharia e Tecnologia, 2010, 2, 1, pp 9-16, 5 ref.

